Apenas para uso em pesquisa. Não utilizar em procedimentos diagnósticos.
The SARS-CoV-2 Lambda variant (C.37) was first detected in Peru in December 2020 and was classified as a Variant of Interest (VOI) by the WHO in June 2021, due to a significant increase in its presence in Peru, Chile, the United States, Argentina, and other countries.
The spike protein of the Lambda variant exhibits higher infectivity, primarily attributed to the T76I and L452Q mutations. Another mutation, F490S, has been associated with reduced susceptibility to antibodies generated by recovered COVID-19 patients.
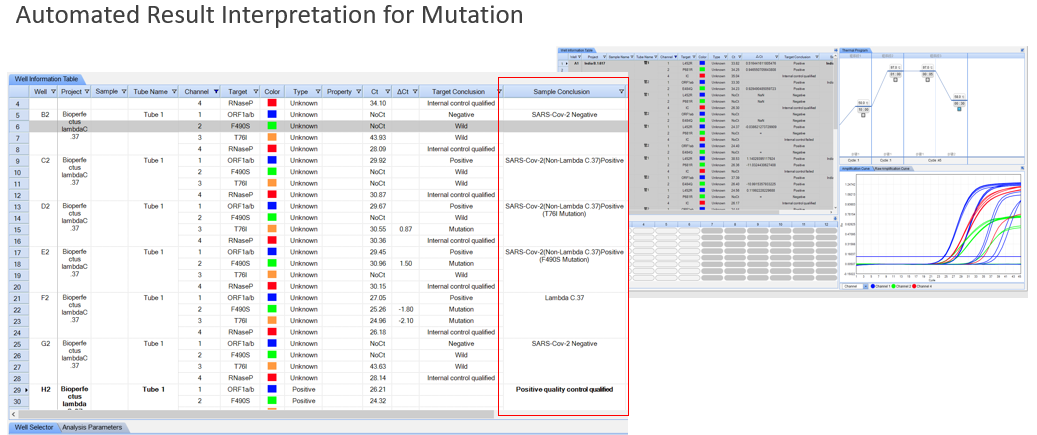
This kit assists in identifying the Lambda variant by simultaneously detecting ORF1a/b, F490S, and T76I, providing fast and practical interpretation through an automated result analysis tool.
Our R&D team closely monitors variants reported globally and develops assays to detect emerging mutations related to increased infectivity, immune escape, and impact on vaccines and drug therapies.
Multiplex: Simultaneous qualitative detection of four targets (ORF1a/b, F490S, T76I, and internal control).
One-tube: One-tube real-time PCR reaction with four colors.
Smart: Automated interpretation of results for STC-96A and STC-96A Plus* instruments.
Efficient: Detection of 94 samples in 72 minutes.
Reliable: Endogenous internal control (IC) ensuring reliability throughout the process.
Versatile: Semi-automatic interpretation tool available for other real-time PCR instruments.
| Targets: |
|
| Positive Percent Agreement | 100% |
| Negative Percent Agreement | 100% |
| Limit of Detection (LoD) | 500 copies/mL |
| Sample types |
Nasopharyngeal swab, throat swab, and sputum samples |
Apenas para uso em pesquisa. Não utilizar em procedimentos diagnósticos.